
MATTHAEUS LABORATORY
Info

In the lab we are interested in a specific organelle found at the plasma membrane and cytosol called caveolae. Our group
combines advanced light and electron microscopy techniques with physiological approaches in vivo.
Research
Project 1: Caveolae in obesity and type 2 diabetes
The uptake of nutrients is a highly regulated process that when misregulated can promote the development of diseases such as type 2 diabetes, cancer or cardiovascular diseases. Previous studies showed that caveolae - 50-80 nm sized plasma membrane invaginations - are essential for cellular lipid uptake in vivo. Loss of caveolae results in impaired lipid uptake and reduced lipid accumulation. In our group we aim to understand how caveolae number and distribution change during extensive weight gain and obesity progression. Previously, we showed that caveolae are more mobile in obese conditions (Matthaeus et al, PNAS 2020) and detach from the plasma membrane regularly (see EM image below). Goal of the project is to determine the spatial distribution of caveolae in metabolic tissues (fat, muscle, liver) during obesity progression, and how caveolae may influence type 2 diabetes development. We therefore use diet induced obesity mouse models and investigate caveolae from gene to function.


Caveolae in lipid metabolism. Left: Scheme illustrates phenotypes differing in caveolae number or plasma membrane stabilization and their influence on lipid accumulation in lipid droplets (made with BioRender). Right: Caveolae are often detached from the plasma membrane in fat cells (adipocytes) of obese mice (ob/ob) compared to control mice (C57BL6/N, black arrows indicate caveolae). EM image taken from Matthaeus et al., PNAS 2020.
Project 2: Caveolae mediated lipid trafficking
Caveolae are mobile organelles that move within the plasma membrane and can get internalized into the cytosol. By using TIRF microscopy we can track single caveolae labelled with fluorescence tags (e.g. caveolin1-eGFP) at the plasma membrane in live cells (see video).
How can caveolae facilitate lipid uptake into cells (and across the plasma membrane)? And what happens next?
Previous studies revealed that caveolae migrate from the plasma membrane to lipid droplets after extracellular lipid treatment (Matthaeus & Taraska, 2021). However, it is currently not understood how caveolae trafficking is realized in detail and how the trafficking routes are regulated in different cell types. In the group, we aim to elucidate the mechanism of caveolae mediated lipid uptake and trafficking by applying advanced light and electron microscopy techniques. Identification of cellular details in healthy cells is key for understanding changes in caveolae lipid trafficking in obesity or type 2 diabetes.

Live cell TIRF imaging of single caveolae visualized by caveolin1-EGFP (in MEFs).

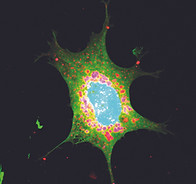
Primary adipocytes stained for lipid droplets (BODIPY, in green), caveolae (caveolin1, in red) and nuclei (DAPI, in blue).
Publications
2025
Esther Ocket, Tamara Pietrucik, Luis Wong Dilworth, Michaela Rath, Daniel Aik, Dmytro Puchkov, Max Ruwolt, Martin Lehmann, Claudia Matthaeus#, Lipid-induced Caveolin1-Lipid droplet trafficking is associated with lipid droplet growth, BioRxiv doi.org/10.64898/2025.12.10.693432
2024
Esther Ocket, Claudia Matthaeus#, Insights in caveolae protein structure arrangements and their local lipid environment, Biological Chemistry, doi.org/10.1515/hsz-2024-0046
R. Groza, K. V. Schmidt, P. M. Müller, P. Ronchi, C. Schlack-Leigers, U. Neu, D. Puchkov, R. Dimova, Claudia Matthaeus, J. Taraska, T. R. Weikl, H. Ewers, Adhesion energy controls lipid binding-mediated endocytosis, Nature Communications, 15, 2767, https://doi.org/10.1038/s41467-024-47109-7
F. Lukas, Claudia Matthaeus, T. López-Hernández, I. Lahmann, N. Schultz, M. Lehmann, D. Puchkov, J. Pielage, V. Hauke, T. Maritzen, Canonical and non-canonical integrin-based adhesions dynamically interconvert. Nature Communications, 15, 2093, https://doi.org/10.1038/s41467-024-46381-x
2023
D. Puchkov, P.M. Müller, M. Lehmann, Claudia Matthaeus, Analyzing the cellular plasma membrane by fast and efficient correlative STED and platinum replica EM, Frontiers in Cell and Developmental Biology, https://doi.org/10.3389/fcell.2023.1305680
X. Li, Y. Wu, Y. Su, I. Rey-Suarez, Claudia Matthaeus, T. B. Updegrove, Z. Wei, L. Zhang, H. Sasaki, Y. Li, M. Guo, J. P. Giannini, H. D. Vishwasrao, J. Chen, S. J. Lee, L Shao, H.Liu, K S. Ramamurthi, J. W. Taraska, A. Upadhyaya, P. La Riviere, H. Shroff, Three-dimensional structured illumination microscopy with enhanced axial resolution, Nature Biotechnology, https://www.nature.com/articles/s41587-022-01651-1
Claudia Matthaeus, R. Juettner, M. Gotthardt, F. G. Rathjen, The IgCAM CAR regulates gap junction mediated coupling on embryonic cardiomyocytes and affects their beating frequency, Life, 13, 1-14, doi.org/10.3390/life13010014
2022
Claudia Matthaeus, K. Sochacki, A. Dickey, D. Puchkov, V. Haucke, M. Lehmann, J. Taraska, The molecular components and organization of flat and curved caveolae indicates bendable structural units at the plasma membrane, Nature Communications, 13, 7234, doi.org/10.1038/s41467-022-34958-3
(predisposed BioRxiv: doi.org/10.1101/2022.03.31.486578 )
2021
C. Fryklund, B. Morén, S. Shah, M. Grossi, E. Degerman, Claudia Matthaeus and K. G. Stenkula, EHD2 deficiency restricts adipose tissue expansion and impairs lipolysis in primary inguinal adipocytes, Frontiers in Physiology, doi.org/10.3389/fphys.2021.740666
Claudia Matthaeus#, J. Taraska, Energy and dynamics of caveolae trafficking, Frontiers in Cell and Developmental Biology, 8:614472, doi.org/10.3389/fcell.2020.614472
2020
Claudia Matthaeus#*, I. Lahmann*, S. Kunz, W. Jonas, A. Melo, M. Lehmann, E. Larsson, R. Lundmark, M. Kern, M. Blüher, H. Olschowski, J. Kompa, B. Brügger, D. N. Müller, V. Haucke, A. Schürmann, C. Birchmeier and O. Daumke, EHD2 mediated restriction of caveolar dynamics regulates cellular fatty acid uptake, PNAS, 117: 7471 – 7481, doi.org/10.1073/pnas.1918415117
G. Fan, M. Kaßmann, Y. Cui, Claudia Matthaeus, S. Kunz, C. Zhong, S. Zhu, Y. Xie, D. Tsvetkov, O. Daumke, Y. Huang, M. Gollasch, Age attenuates the T-type CaV3.2-RyR axis in vascular smooth muscle, Aging Cell, 00:e13134, doi.org/10.1111/acel.13134
2019
Claudia Matthaeus#, X. Lian, S. Kunz, M. Lehmann, C. Zhong, C. Bernert, I. Lahmann, D. N. Müller, M. Gollasch and O. Daumke, eNOS-NO induced relaxation of small blood vessels requires EHD2-dependent stabilization of caveolae at the plasma membrane, Plos One, 14(10): e0223620, doi.org/10.1371/journal.pone.0223620
X. Lian, Claudia Matthaeus, M. Kaßmann, O. Daumke, M. Gollasch. Pathophysiological Role of Caveolae in Hypertension, Frontiers of Medicine, 6:153, doi.org/10.3389/fmed.2019.00153
K. Faelber, L. Dietrich, J. K. Noel, F. Wollweber, A. Pfitzner, A. Mühleip, R. Sánchez, M. Kudryashev, N. Chiaruttini, H. Lilie, J. Schlegel, E. Rosenbaum, M. Hessenberger, Claudia Matthaeus, S. Kunz, A. von der Malsburg, F. Noé, A. Roux, M. van der Laan, W. Kühlbrandt and O. Daumke. Structure and assembly of the mitochondrial membrane remodeling GTPase Mgm1, Nature, 571: 429-433, doi.org/10.1038/s41586-019-1372-3
2017
Claudia Matthäus#, H. Langhorst, L. Schütz, R. Jüttner and F. G. Rathjen, Cell-cell communication mediated by the CAR subgroup of immunoglobulin cell adhesion molecules in health and disease, Molecular and Cellular Neuroscience, 81: 32-40, doi.org/10.1016/j.mcn.2016.11.009
2015
Claudia Matthäus, The IgCAM CAR regulates calcium homeostasis in the developing heart, PhD Thesis, https://refubium.fu-berlin.de/handle/fub188/2888
2014
Claudia Matthäus#, J. Schreiber, R. Jüttner and F. G. Rathjen, The IgCAM CAR is implicated in cardiac development and modulates electrical conduction in the mature heart. Journal of Cardiovascular Development and Disease, 1: 111-120, doi.org/10.3390/jcdd1010111
U. Hempel, Claudia Matthäus, C. Preissler, S. Möller, V. Hintze, P. Dieter, Artificial Matrices With High-Sulfated Glycosaminoglycans and Collagen Are Anti- Inflammatory and Pro-Osteogenic for Human Mesenchymal Stromal Cells. Journal of Cellular Biochemistry, 115: 1561-1571, doi.org/10.1002/jcb.24814
Team

Luis Wong Dilworth, PhD
I joined the Matthaeus Lab in October 2024 as a postdoc after completing my PhD at the Free University of Berlin, where I studied the role of ARF GTPases in ER-Golgi transport using STED super-resolution microscopy. My research now focuses on understanding how caveolae machinery drives lipid trafficking at the plasma membrane in caveolae-lacking cells, combining advanced light and electron microscopy with CRISPR-Cas9 gene editing
Contact
claudia.matthaeus(at)uni-potsdam.de
Institute for Nutritional Science
University of Potsdam
Follow on Bluesky: @cmatthaeus.bsky.social





